Covid-19 Antigen Test
A COVID-19 antigen test, also recognized as a rapid antigen test or rapid antigen detection test (RADT), constitutes in vitro diagnosis used to detect the presence of viral antigens associated with the SARS-CoV-2 virus, which cause COVID-19 infection.
Healthcare providers commonly use antigen tests to screen and diagnose , especially in situations requiring swift outcomes.
The COVID-19 antigen test functions by detecting distinct proteins from the SARS-CoV-2 virus in respiratory samples, like nasal or throat swabs. These tests are recognized for their speedy results, often available in 15 to 30 minutes. Their value lies in swiftly pinpointing potentially infectious individuals, enabling prompt implementation of public health actions such as isolation and contact tracing.
What is Covid-19 Antigen Test ?
While antigen tests are rapid and convenient, Covid-19 antigen test it’s important to note that they may carry a slightly higher potential for generating false-negative results compared to molecular tests such as polymerase chain reaction (PCR) tests.
Healthcare providers utilize the COVID-19 AT as a diagnostic tool to identify the presence of the SARS-CoV-2 virus, the causative agent of COVID-19. This test detects viral proteins (antigens) in respiratory samples, typically acquired from the nasal cavity using a swab. The following presents information about COVID-19 AT:
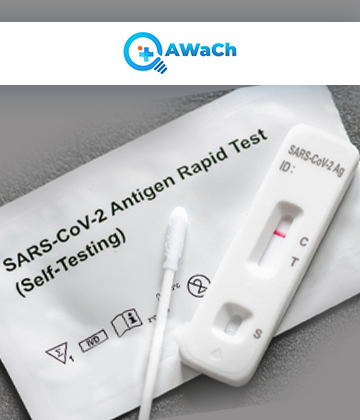
A COVID-19 Antigen Test, often referred to as a rapid antigen test or rapid antigen detection test (RADT), represents an invaluable in vitro diagnostic tool used for detecting the presence of viral antigens linked to the SARS-CoV-2 virus, responsible for causing COVID-19 infection.
Healthcare providers extensively employ antigen tests to swiftly screen and diagnose COVID-19, particularly in scenarios where rapid results are imperative. The mechanism of the COVID-19 antigen test involves the detection of specific proteins derived from the SARS-CoV-2 virus in respiratory specimens, including nasal or throat swabs. These tests are renowned for their expeditious turnaround time, frequently delivering results within 15 to 30 minutes. Their significance lies in the prompt identification of potentially contagious individuals, facilitating immediate implementation of crucial public health measures such as isolation and contact tracing.
Strength:
The strength of COVID-19 Antigen Tests lies in their ability to provide rapid and on-the-spot results, making them invaluable in situations requiring swift action to control the spread of the virus. These tests offer a pragmatic approach to mass screening, allowing healthcare professionals to quickly identify and isolate infected individuals, thereby curbing transmission. The convenience and speed of antigen tests are particularly advantageous in settings like airports, schools, and healthcare facilities, where timely decision-making is vital.
Advantages:
COVID-19 Antigen Tests offer several advantages, including their rapidity, accessibility, and cost-effectiveness. Their ability to provide results within minutes enables timely intervention, reducing the risk of further transmission. While molecular tests like polymerase chain reaction (PCR) tests are highly accurate, antigen tests provide a valuable initial screening tool. It's crucial to note that while antigen tests are generally reliable, they may have a slightly higher risk of generating false-negative results compared to PCR tests. Therefore, healthcare professionals often use clinical judgment and consider additional testing if there is a strong suspicion of COVID-19 despite a negative antigen test result.
Recognitions:
COVID-19 Antigen Tests have gained recognition and widespread use in the global fight against the COVID-19 pandemic. They have been instrumental in expanding testing capacity and enabling rapid decision-making in various healthcare and public health settings. The development of high-sensitivity tests like InstaCOVID Ag by QAWaCh Bio reflects ongoing innovation in this field, further enhancing the utility of antigen testing.
Innovation:
QAWaCh Bio's InstaCOVID Ag represents a notable innovation in the realm of COVID-19 Antigen Tests. This ICMR and CDSCO-approved test kit utilizes QAWaCh Bio's proprietary signaling molecules 'Q-Probe' to achieve ultrahigh sensitivity. It delivers test results within an astonishingly short 10-minute timeframe, offering a game-changing solution for point-of-care testing. Such innovations exemplify the commitment of the medical and scientific community to enhancing the accuracy and accessibility of COVID-19 testing, ultimately contributing to the global effort to combat the pandemic.
